At Boston Scientific, we have Deep Brain Stimulation (DBS) options to fit your life. Your specialist care team will take you through the best options suited to your needs.
Types of Implantable Pulse Generators (IPGs)
There are two types of devices available:
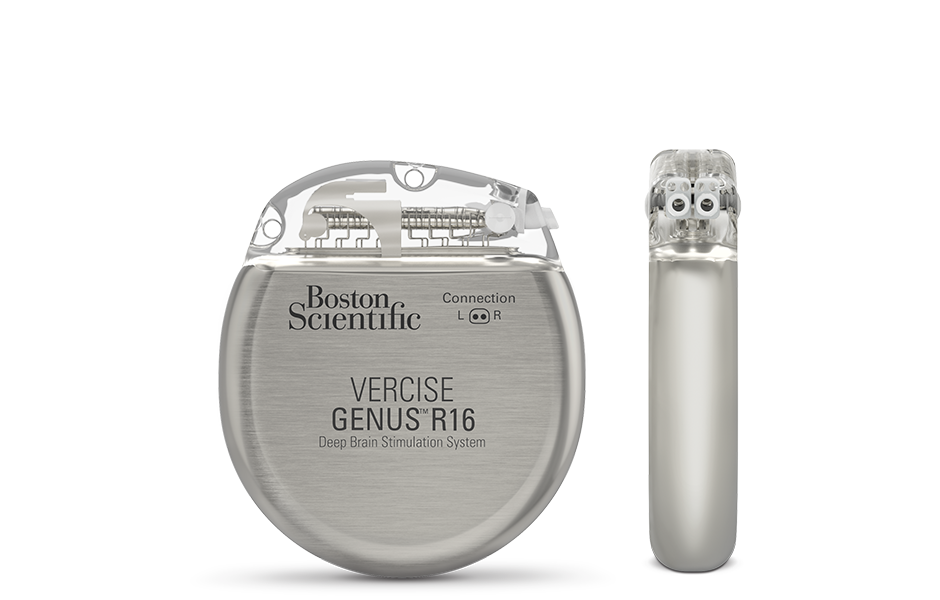 Vercise Genus™ R16 | Rechargeable - Lasts at least 25 years* - To avoid future surgeries.
Vercise Genus™ R16 | Rechargeable - Lasts at least 25 years* - To avoid future surgeries.
 Vercise Genus™ P16 | Non-rechargeable - Lasts about 3–5 years* - The thinnest dual-channel device available.
Vercise Genus™ P16 | Non-rechargeable - Lasts about 3–5 years* - The thinnest dual-channel device available.
What is the difference?
*Battery life is dependent on the stimulation settings and conditions.
What makes Boston Scientific DBS systems unique?
As you think about your DBS options, it is important to consider the programming of the device. The programming of the device – how your therapy is set-up, delivered, and maintained over time – ensures you are receiving therapy that meets your needs.
Typically, DBS programming is perfected over time. Getting to a point in the therapy that balances symptoms and side-effects can take some time and is achieved over many programming sessions.
We have accepted the challenge to improve this process. Our solution: Image Guided DBS provides your physician the ability to see exactly where your leads are located and exactly where your stimulation needs to be delivered.
We hear often – How do you know the leads are in the right place? Or if my condition changes, can my therapy change too? Image Guided DBS answers both questions.
See how Image Guided DBS works:
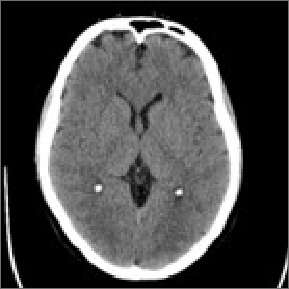
Images are collected during your DBS procedure.
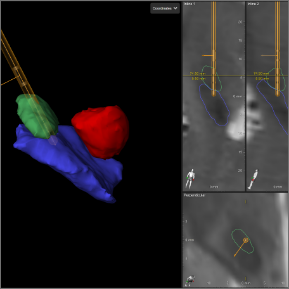
Collected images are evaluated to see the exact location of your leads.
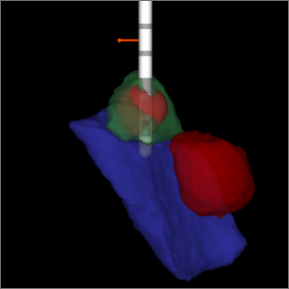
The images are loaded into the programming software and therapy is programmed precisely – based on the location of your leads.

Stimulation can be adjusted to minimize side affects, at the initial programming and in the future.
Giving you your time back.
When your device is programmed with Image Guided DBS you may spend less time in your programming sessions.** A recent study of 10 patients showed an initial programming time of 20 minutes; this is less than half the time typically spent in a session. The study concluded that Image Guided Programming drastically reduces programming time without compromising symptom control and patient satisfaction.¹


This material is for informational Purposes only and not meant for medical diagnosis. This information does not constitute medical or legal advice, and Boston Scientific makes no representation regarding the medical benefits included in this information. Boston Scientific strongly recommends that you consult with your physician on all matters pertaining to your health.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.
References:
** Results from case studies are not necessarily predictive of results in other case studies. Results in other studies may vary.
1. Lange F, 2021, Front. Neurol. doi: 10.3389/FNEUR.2021.785529/BIBTEX